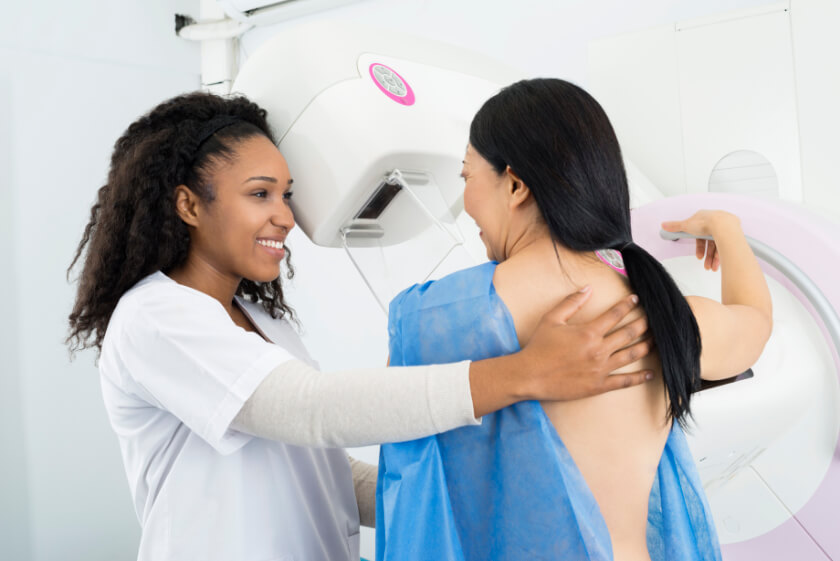
FDA Relaxes Guidelines for Mammography During Coronavirus

The COVID-19 pandemic has complicated nearly every aspect of daily life, especially for healthcare professionals and the facilities where they work. Many clinics are overwhelmed with patients; others have had to cut back on the number of workers they employ. It has also complicated care for patients who need diagnostic and screening tests, such as mammograms. Furthermore, the pandemic has made it tougher for healthcare facilities to fulfill some of the regulations required to operate.
The U.S. Food and Drug Administration (FDA) plays a critical role in helping patients get the quality care they need by implementing certain regulations on healthcare providers. Sometimes, though, these regulations get in the way of healthcare; this is especially true during the coronavirus health emergency. To help both patients and healthcare providers, the FDA issued guidance to help imaging facilities provide mammography services to their patients in a safe way during the pandemic.
About the Role the FDA Plays in Mammography
The FDA sets quality standards for specific procedures, such as mammography, to make sure all patients receive high quality care. The Mammography Quality Standards Act of 1992 (MQSA) requires mammography facilities undergo accreditation, certification and inspection of equipment and personnel, for example. Certification means the FDA or a state certification board has certified that a mammography facility is capable of providing a quality mammography. To receive certification, the mammography provider must have specific mammography equipment, for example, and undergo inspection from FDA or State inspectors that have received special training in certifying mammography equipment.
Certified mammography facilities must also put their mammography equipment through annual testing, known as a survey, and submit a survey report to the FDA. The survey report contains information, such as how much radiation the equipment uses to create the images, the intensity of the x-ray beam, and the accuracy and precision of the mammogram images. The facility must also undergo accreditation, which is a rigorous review process that requires a substantial amount of labor, both on the part of the facility and the FDA.
FDA Issues Temporary Guidance for Mammography during Coronavirus
The Secretary of Health and Human Services (HHS) declared a public health emergency in January of 2020. In the months that followed, it became clear that coronavirus was slowing down the smooth delivery of healthcare in the nation. Concerns about overwhelmed hospitals, limited supplies, and patients delaying routine screenings and procedures caused job losses among healthcare professionals, which then left many clinics and other facilities short-staffed. Workers that remained continued to provide outstanding healthcare services, of course, but many struggled to meet certain regulatory standards. In other words, they had trouble keeping up with paperwork.
The FDA took action to help healthcare professionals focus on providing high quality healthcare during coronavirus, instead of getting tangled up in paperwork. On December 4, 2020, the FDA issued the Enforcement Policy for the Quality Standards of the Mammography Quality Standards Act During the COVID-19 Public Health Emergency, which took effect immediately. The FDA’s policy will remain in effect only for the duration of the pandemic.
The FDA’s guidance helps facilities that temporarily stopped performing mammography during the pandemic. It also helps those facilities that are continuing to perform mammography but that are having difficulty complying with certain quality standard requirements because of the public health emergency.
The FDA will grant some leeway to mammography centers that cannot meet the normal requirements detailed in the Mammography Quality Standards Act. By relaxing certain restrictions, the FDA hopes to increase the availability of mammography during the ongoing coronavirus pandemic.
Travel restrictions, quarantine, and the closure of some healthcare centers have prevented some mammography providers from scheduling the annual on-site surveys required under MQSA. This could potentially cause a large backlog of scheduled surveys once the pandemic has ended. To avoid this backlog, the FDA said it would not object if mammography providers complete their surveys after the set MQSA deadlines, but the administration does encourage facilities to complete their surveys as soon as possible.
Normally, mammography facilities have to complete the survey within 14 months of their original survey – the FDA would like these clinics to submit their surveys within 6 months of that 14-month anniversary. Under the temporary guidelines, the mammography facilities would also have to provide documented information regarding the delay. Facilities that have not had an annual survey for 20 months should call the MQSA hotline; the FDA will review them on a case-by-case basis. The FDA also offered guidance to mammography providers who are struggling to meet other MQSA requirements.
In short, the FDA said that it would not object when mammography facilities that have experienced difficulties in complying with certain standards in cases in which the mammography providers had no control over the non-compliance, and when non-compliance does not cause undue risk to patient safety or the quality of the mammograms. In other words, the FDA loosened guidelines on paperwork to help doctors provide the mammography patients need to stay healthy during the pandemic.